 Download PDF
Download PDF
Name: Cuscuta cephalanthi Engelm.
Family: Convolvulaceae, the Morning Glory Family
Common Names: Buttonbush dodder, butterbush dodder (4,23).
Etymology: With Arabic origins, Kushkut, means dodder plant or parasitic plant; in New Latin, Cuscuta directly translates as dodder. In Greek, cephalo converts to head and cephalus to mullet, a type of fish. Anthos directly translates to flower in Greek, yet anthus translates to wagtail, the bird, in Latin. The genus Cephalanthus is the buttonbush, so named for the round clusters of flowers looking like round buttons. The species epithet, cephalanthi refers to the tendency of the plant to parasitize Cephalanthus (1,4).
 Botanical synonyms (2):
Botanical synonyms (2):
Epithymum cephalanthi (Engelm.) Nieuwl. & Lunell
Grammica cephalanthi (Engelm.) W.A. Weber
Quick Notable Features:
¬ Reduced leaves and very thin stems (L.S.S. pers. obs.)
¬ String-like stem is 0.4-0.6mm in diameter (17)
¬ Tiny flowers, ~2-4mm, with translucent yellowish-white petals, growing in spicate-paniculate clusters; either sessile or short-pedicelled (1,8,16,17)
¬ Perianth lobes are obtuse, most often 4-merous, not papillate and exhibit pellucid gland dots (4,8,16,18,20)
¬ Apparent corolla tube (L.S.S. pers. obs. of botanical illustrations in 4,24)
¬ Often present on, but not restricted to Cephalanthus (1,14,17)
Plant Height: The height of Cuscuta cephalanthi is dependent on the host; H.L. Dean measured the length of a single dodder plant at nearly half a mile (19).
Subspecies/varieties recognized: None found in the literature.
Most Likely Confused with: Any other species of Cuscuta—in Michigan these might be: C. coryli, C. epilinum, C. epithymum, C. glomerata, C. indecora, C. pentagona or C. polygonorum.
Habitat Preference: In Michigan, C. cephalanthi parasitizes Amphicarpaea, Boehmeria, Cephalanthus, Equisetum, Lycopus, Lythrum, Populus, Salix, Sambucus, Spiraea, Stachys and many Compositae (e.g., Achillea, Aster, Cirsium, Eupatorium and Solidago). It also inhabits the periphery of water bodies: streams, rivers, ponds, lakes, and swamps (1,2,16).
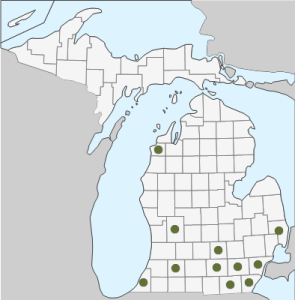
Geographic Distribution in Michigan: C. cephalanthi is found in 11 counties in Michigan’s Lower Peninsula: Berrien, Benzie, Kalamazoo, Kent, Jackson, Ingham, Lenawee, Washtenaw, Monroe, Wayne and St. Clair (1).
Known Elevational Distribution: The altitudinal range of C. cephalanthi is between 0 and 3,000m in California (5).
Complete Geographic Distribution: C. cephalanthi is native to the continental U.S. and Canada. It is found throughout the United States, excluding the mid-west (Colorado, Wyoming, Montana), southeast (Louisiana, Mississippi, Alabama, and Florida), and a few states in the east (West Virginia, New Jersey, and Delaware) and the northeast (Vermont and New Hampshire). It is present in the following Canadian provinces: British Columbia, Alberta, Manitoba, Ontario, Nova Scotia, and New Brunswick (6).
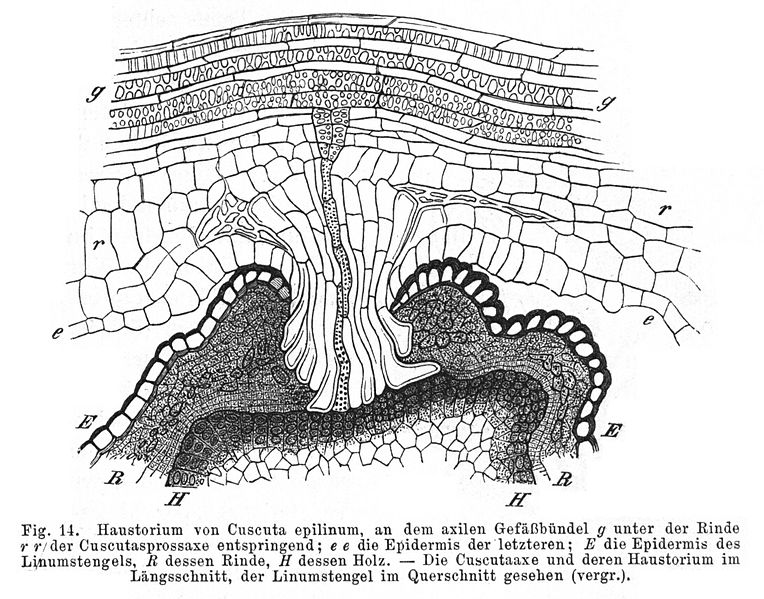 Parasitism: Parasitism is a type of symbiotic relationship in which one organism obtains nutrients directly from a host organism. This has a detrimental effect on the host, but benefits the parasite. Although parasitic plants are commonly known to lack chlorophyll, some species have green organs, making them partially photoautotrophic. The physical link between the parasite and the host is called a “haustorium,” and often occurs through xylem-to-xylem attachment. The host can vary, ranging from the mycorrhizae of trees, to grasses and hardwood trees. The parasite often maintains open or partially open stomata, allowing transpiration to aid in extracting nutrients from the host (11). For further reading on haustoria see (22): The Physiology of Plants Under Stress.
Parasitism: Parasitism is a type of symbiotic relationship in which one organism obtains nutrients directly from a host organism. This has a detrimental effect on the host, but benefits the parasite. Although parasitic plants are commonly known to lack chlorophyll, some species have green organs, making them partially photoautotrophic. The physical link between the parasite and the host is called a “haustorium,” and often occurs through xylem-to-xylem attachment. The host can vary, ranging from the mycorrhizae of trees, to grasses and hardwood trees. The parasite often maintains open or partially open stomata, allowing transpiration to aid in extracting nutrients from the host (11). For further reading on haustoria see (22): The Physiology of Plants Under Stress.
Vegetative Plant Description: When individuals of Cuscuta germinate, they develop a short anchorage root, and a stem emerges from the seed, nutating (rotates) in search of a host. When an attachment with a host has been created, the anchorage root dies (7). Additional means of finding a host have been suggested in literature, such as positive phototropism or growth toward a source of moisture or specific chemicals (12). In C. cephalanthi, the stems are simple, leaves are extremely reduced to absent (alternate if present); the stems are string-like (0.4-0.6mm diameter) and cream-yellow-green (17).
Climbing Mechanism: In the genus Cuscuta, two types of coiling methods are found: loosely twining with few haustoria, and tightly twining with many haustoria—both show upward growth only; it is suggested that the former is used while the plant is in search of a host and the latter is used once contact with a host has been made (7). Typically, Cuscuta spp. makes no more than three turns around the same branch. (12). Kuijt states that the genus generally twines dextrally (7).
Flower Description: The flower is tiny, measuring ~2-4mm, with translucent yellow-white petals; they originate endogenously from a part of the stem called the haustorial coil (7,22), growing in cymose spicate-paniculate clusters and are sessile or short-pedicelled, ranging from 0-1.2mm. The sepals are connate, 1-2mm long, most often 4-merous, but can be 3- and 5-merous and is usually shorter than the corolla tube; its lobes are ovate and can exhibit irregular margins. The 2-4mm perianth lobes are ovate, obtuse, erect to spreading, shorter than the corolla tube, not papillate and exhibit pellucid gland dots. The corolla tube is cylindric-campanulate and as it matures, it expands to cover most of the capsule. The corolla is, usually 4-parted and less commonly 3- or 5-parted (4,8,16,17,18,20). The two styles (1-1.5mm long) are narrow and equal to or longer than the ovary, which is depressed globose-ovoid, bilocular, and 0.9-2mm; the stigmas are globose-capitate. The stamens are equal in length or shorter than the corolla lobes; the anthers are ovoid to spherical and the filaments are subulate; the anthers (0.4-0.7mm) and filaments (0.2-0.6mm) are about equal in length. The presence of infra-staminal scales is important to recognize—they bear scattered processes and are oblong, nearly reaching the height of the filaments (1,8,16,17,20).
Flowering Time: In Ohio and the Northeast, C. cephalanthi flowers from August to September (4,15). In Flora of the Great Plains, the flowering period is listed as July-October (17).
Pollinator: Yuncker observed visits by wasps and other species of the order Hymenoptera to Cuscuta (8). Specific pollination information on C. cephalanthi was not found in the literature.
 Fruit Type and Description: The fruit is a capsule 2-2.5mm in diameter, initially globose to ovoid, but later developing as depressed-globose; the corolla tube can persist around the capsule, with lobes at the summit, or fall off entirely (8,17,21).
Fruit Type and Description: The fruit is a capsule 2-2.5mm in diameter, initially globose to ovoid, but later developing as depressed-globose; the corolla tube can persist around the capsule, with lobes at the summit, or fall off entirely (8,17,21).
Seed Description: In the genus Cuscuta, the defining characteristic of the mature embryo is the absence of cotyledons. This may be attributable to the fact that the first job of the young stem is to search for a host, not to photosynthesize. Each ovary bears four ovules, but one or more may abort, which causes variation in seed size and shape; a Cuscuta seed may have zero, one, or two flat surfaces (7). In C. cephalanthi, the seed is 1.5-2mm long, with an outline shape varying between oval-ovoid to orbiculate; it is compressed and slightly oblique (8,17).
Dispersal Syndrome: C. cephalanthi is often seen growing on woody and herbaceous hosts, particularly Cephalanthus occidentalis, so the presence of the host is a factor in its dispersal (17). In unspecified members of the genus Cuscuta, both germination in the capsule and seeds falling to the ground were observed, leaving water dispersal or other means a possibility for dispersal. Additionally, Cuscuta spp. seeds may be able to pass through the intestinal tract of a sheep intact, remaining viable. Although this method of dispersal is unlikely, it extends the potential dispersal mechanisms to include zoochory (7).
Distinguished by: Close similarities between the vegetative parts of species of dodders leave one to rely on floral, fruit, and inflorescence characteristics to distinguish among the species (7). C. cephalanthi may be most closely related to C. coryli and C. polygonorum. The confusion point is that each of these species is usually 4-merous. C. cephalanthi can be distinguished by its obtuse to rounded lobes and styles that are approximately as long as the capsule. C. coryli contrasts with its acute, incurved lobes and styles that are shorter than the capsule. Further, some to all of the flowers in C. coryli are pedicellate and the perianth is papillate (1,15). C. polygonorum is differentiated from the above two species by its erect or ascending corolla lobes (15).
Other members of the family in Michigan (number species): In Michigan, Convolvulaceae is a small family, comprised of four genera: Calystegia (3), Convolvulus (1), Ipomoea (4), and Cuscuta with 10 species: C. campestris Yuncker, C. coryli Engelm., C. epilinum Weihe, C. epithymum (L.) L., C. glomerata Choisy, C. gronovii Schultes, C. indecora Choisy, C. pentagona Engelm. and C. polygonorum Engelm. (1).
Ethnobotanical Uses: While no specific information has been located on the use of this species of Cuscuta, the following information that is not attributed to any one species has been found:
“An Indian proverb states that the person finding the root of dodder will have access to all the riches of the earth” (7). This statement pertains to the wide use of Cuscuta spp. for medicinal purposes across Asia, from herbal mixtures to treat ovarian cancer and postmenopausal osteoporosis to antifungal and insecticidal applications (10).
 Phylogenetic Information: Convolvulaceae is among five other families of the order Solanales (Montiniaceae, Sphenocleaceae, Hydroleaceae, and Solanaceae), which encompasses 165 genera and 4,080 species. The distribution of Convolvulaceae is extensive worldwide, excluding areas of extreme temperatures—the Sahara and Gobi Deserts, and areas of high latitude (Canada, Greenland, Russia, Antarctica, as well as the southern tip of South America). Convolvulaceae has been noted as the only asterid family whose seeds exhibit physical dormancy (7). Cuscuta spp., belonging to the subfamily Cuscutoideae, is the only genus within the family that is parasitic. Its placement in Convolvulaceae is openly debated, but is supported by similar flower morphology (7,8,9) as well as the twining habit.
Phylogenetic Information: Convolvulaceae is among five other families of the order Solanales (Montiniaceae, Sphenocleaceae, Hydroleaceae, and Solanaceae), which encompasses 165 genera and 4,080 species. The distribution of Convolvulaceae is extensive worldwide, excluding areas of extreme temperatures—the Sahara and Gobi Deserts, and areas of high latitude (Canada, Greenland, Russia, Antarctica, as well as the southern tip of South America). Convolvulaceae has been noted as the only asterid family whose seeds exhibit physical dormancy (7). Cuscuta spp., belonging to the subfamily Cuscutoideae, is the only genus within the family that is parasitic. Its placement in Convolvulaceae is openly debated, but is supported by similar flower morphology (7,8,9) as well as the twining habit.
 Interesting Quotation or Other Interesting Factoid not inserted above:
Interesting Quotation or Other Interesting Factoid not inserted above:
Cuscuta cephalanthi, along with six other Cuscuta species, were originally published by George Engelmann in 1842 (13). Thereafter, he discovered that many Cuscuta spp. are not specific to one host, including C. cephalanthi to Cephalanthus. In a subsequent article he stated his wish to change the name to C. tenuiflora (14); after much discussion with Dr. Asa Gray, the names were left as is. C. tenuiflora was never published as a new name (avowed substitute, nomen novum), therefore C. tenuiflora is illegitimate (nomen superfluum). Nancy Hensold of the Field Museum kindly helped the first author interpret this information. The original description of C. cephalanthi is tremendous; do take a look at it online via the Biodiversity Heritage Library.
Reflecting on how the growth of Cuscuta impacts cultures: “The dodder’s rapid development and its stranglehold on and damage to the host have earned it a place in the superstition of many Western countries. The German “Teufelsxwirn” and Dutch “Duivelsnaaigaren” are vernacular names of this sort,” highlights Cuscuta’s standing as a noxious weed in many places (7).
Literature and websites used:
- Voss, E.G. 2004. Michigan Flora Part III: Dicots Concluded. Ann Arbor, MI: Cranbrook Institute of Science.
- Tropicos.org. Missouri Botanical Garden. 13 Apr 2012 <http://www.tropicos.org/Name/8500666>
- Brown, R.W. 1956. Composition of Scientific Words. Washington, D.C., USA: Smithsonian Institution Press.
- Cooperrider, T.S. 1995. The Dicotyledons of Ohio: Linaceae through Campanulaceae. Columbus, OH, USA: Ohio State University Press.
- The Calflora Database 2012. Berkeley, CA. http://www.calflora.org/cgi bin/species_query.cgi?where-calrecnum=2533 (Accessed: April 16, 2012).
- USDA, NRCS. 2008. The PLANTS Database, Version 3.1, National Plant Data Center, Baton Rouge, LA 70874-4490 USA. http://plants.usda.gov/java/profile?symbol=cuce
- Kuijt, J. 1969. The Biology of Parasitic Flowering Plants. Los Angeles, CA, USA: University of California Press
- Yuncker, T.G. 1920. Revision of the North American and West Indian Species of Cuscuta. Illinois Botanical Monographs 6(2&3): 1-141.
- Olmstead, R.G. & S. Stefanović 2004. Testing the phylogenetic position of a parasitic plant (Cuscuta, Convolvulaceae, Asteridae): Bayesian inference and the parametric bootstrap on data drawn from three genomes. Systematic Biology 53(3): 384-399.
- Costea, M. & F.J. Tardif 2006. The biology of Canadian weeds. 133. Cuscuta campestris Yuncker, C. gronovii Willd. ex Schult., C. umbrosa Beyr. ex Hook., C. epithymum (L.) L. and C. epilinum Weihe. Canadian Journal of Plant Science 86(1): 293-316.
- Clark, W.D., R. Moore, & K.R. Stern. 1995. Botany. Dubuque, IA, USA: Wm. C. Brown Publishers.
- Menninger, E.A. 1970. Flowering Vines of the World. New York, NY, USA: Hearthside Press Incorporated.
- Engelmann, G. 1842. Monography of the North American Cuscutineae. The American Journal of Science 43: 336-337.
- Engelmann, G. 1843. North American Cuscutineae. The London Journal of Botany 2: 196- 197.
- Magee, D.W. & H.E. Ahles. 1999. Flora of the Northeast: A Manual of the Vascular Flora of New England and Adjacent New York. Amherst, MA, USA: University of Massachusetts Press.
- Hickman, J.C. 1993. The Jepson Manual of Higher Plants in California. Berkeley, CA, USA: University of California Press.
- McGregor, R.L. 1986. Flora of the Great Plains. Lawrence, KS, USA: The University Press of Kansas.
- Gleason, H.A. & A. Cronquist 1991. Manual of Vascular Plants of the Northeastern United States and Adjacent Canada. Bronx, NY, USA: New York Botanical Garden Press.
- Dean, H.L. 1942. Total length of stem developed from a single seedling of Cuscuta. Proc. Iowa Acad. Sci. 49: 127-128.
- Fernald, M.L. 1950. Gray’s Manual of Botany, 8th ed. NY, USA: American Book Company.
- Radford, A.E., H.E. Ahles, & C.R. Bell 1968. Manual of the Vascular Flora of the Carolinas Chapel Hill, NC, USA: The University of North Carolina Press.
- Orcutt, D.M., E.T. Nilsen, & M.G. Hale 2000. The Physiology of Plants Under Stress, Hoboken, NJ, USA: John Wiley & Sons, Inc.: 443-445.
- ITIS: Integrated Taxonomic Information System http://www.itis.gov/index.html Retrieved 04/13/2012, from the Integrated Taxonomic Information System on-line database.
- Gleason, H. A. 1963. Illustrated Flora of the Northeastern United States and Adjacent Canada, Volume 2. New York, New York, USA: Hafner Publishing Company, Inc.
Image Credits (all used with permission):
http://www.parasiticplants.siu.edu/Cuscutaceae/index.html led to Images 1,3, and 4:
1) Photo by Lytton John Musselman. Gates Co., North Carolina.
2) Drawing of haustoria: Haustorium von Cuscuta epilinum an Linum usitatissimum from Julius Sachs: Vorlesungen über Pflanzenphysiologie, zweite Auflage, Leipzig (1887), downloaded from http://commons.wikimedia.org/wiki/File:Haustorium_Cuscuta_epilinum.jpg
3) Photo by Joel McNeal. Plant cultivated on Coleus in the Pennsylvania State University greenhouse.
4) Photo by Joel McNeal. Plant cultivated on Coleus in the Pennsylvania State University greenhouse.
5) Species distribution map, derived from the Michigan Flora Online.
Primary Authors: Lauren S. Sopher and John Bradtke, with editing by Cristine V. Santanna and Robyn J. Burnham
© Robyn J. Burnham, University of Michigan
For additional information on Michigan Plant Diversity web pages please contact Robyn J. Burnham via email: rburnham“at”umich.edu